General overview
Various species of Cryptosporidium infect most vertebrates. C. parvum infects cattle but can also infect humans; C. hominis appears to be restricted to humans, and began to be recognized in the early 2000s (Hunter 2005) . The oocysts are the infective stage and are about 5 microns in size; they are excreted in feces and are transmitted to new hosts by the fecal-oral route. They are highly resistant to chlorine, but are vulnerable to ultraviolet light disinfection (AWWA 1999) . The durability and infectiousness of the oocysts, as well as their documented ability to cause large outbreaks (Mac Kenzie et al., 1994) , means that control of Cryptosporidium is very important for drinking water treatment. Water treatment utilities should consider all surface water to be contaminated with oocysts (AWWA 1999) . Effective control of Cryptosporidium is generally achieved in drinking water treatment through filtration yielding nonturbid water (<= 0.1 nephelometric turbidity unit) (AWWA 1999).
Cryptosporidiosis is a disease described by a self-limited watery diarrhea with an incubation period of 3 to 7 days (Miliotis & Bier 2003) . Asymptomatic infections are also common in apparently healthy children and adults (Blaser 2002) . However, the disease is particularly dangerous to people with HIV/AIDS because there is no effective treatment (Miliotis & Bier 2003) . This can lead to lethal infections, or chronic disease lasting months or years that severely damages the gut.
Summary of Data
An experiment (DuPont et al., 1995) from feeding an isolate from a calf (Iowa isolate) to human volunteers yields an exponential model with an ID50 of 165 oocysts (Teunis 1999) also fitted a model to these data which is similar to the model presented here.
Messner et al. (2001) described dose response model fits using the complete unpublished data set from DuPont et al., 1995 :
- Reanalysis of stool samples from the above experiment (DuPont et al., 1995) using flow cytometry revealed that 2 individuals thought to be uninfected were actually infected (Messner 2001) .
- Two other feeding studies (Okhuysen et al., 1999) in human volunteers using different isolates yielded models with ID50s of 179 oocysts (UCP isolate, also from a calf) and 9 oocysts (TAMU isolate, from an infected veterinary student).
Messner et al. (2001) fit the exponential model to these three datasets. This was appropriate for the Iowa and TAMU isolates, but the Beta-Poisson model provided a better fit than the exponential model for the UCP isolate. The UCP and TAMU datasets are smaller than the Iowa datasets.
Okhuysen et al. (2002) also conducted a feeding study in adult humans using C. parvum originating from red deer (Moredun isolate).
Chappell et al. (1999) also conducted a feeding study in humans using the Iowa isolate of C. parvum. Although the data were not published, they estimated an ID50 of 83 oocysts for volunteers lacking anti-C. parvum IgG, and an ID50 of 1,880 oocysts for volunteers who had anti-C. parvum IgG.
| ID | Exposure Route | # of Doses | Agent Strain | Dose Units | Host type | Μodel | LD50/ID50 | Optimized parameters | Response type | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| 108 | oral | 8.00 | Iowa strain | oocysts | human | exponential | 1.65E+02 | k = 4.19E-03 | infection |
DuPont, H. L., Chappell, C. L., Sterling, C. R., Okhuysen, P. C., Rose, J. B., & Jakubowski, W. . (1995). The infectivity of Cryptosporidium parvum in healthy volunteers. The New England Journal of Medicine, 332, 13. Retrieved from https://www.nejm.org/doi/full/10.1056/NEJM199503303321304 |
| 139 | oral | 8.00 | Iowa isolate | oocysts | human | exponential | 1.32E+02 | k = 5.26E-03 | infection |
Messner, M. J., Chappell, C. L., & Okhuysen, P. C. (2001). Risk Assessment for Cryptosporidium: A Hierarchical Bayesian Analysis of Human Dose Response Data. Water Research, 35, 16. Retrieved from https://www.sciencedirect.com/science/article/pii/S0043135401001191 |
| 140 | oral | 4.00 | TAMU isolate | oocysts | human | exponential | 1.21E+01 | k = 5.72E-02 | infection |
Messner, M. J., Chappell, C. L., & Okhuysen, P. C. (2001). Risk Assessment for Cryptosporidium: A Hierarchical Bayesian Analysis of Human Dose Response Data. Water Research, 35, 16. Retrieved from https://www.sciencedirect.com/science/article/pii/S0043135401001191 |
| 141 | oral | 4.00 | UCP isolate | oocysts | human | beta-Poisson | 1.79E+02 | a = 1.45E-01 N50 = 1.79E+02 | infection |
Coster, T. S., Wolf, M. K., Hall, E. R., Cassels, F. J., Taylor, D. N., Liu, C. T., … McQueen, C. E. (2007). Immune response, ciprofloxacin activity, and gender differences after human experimental challenge by two strains of enterotoxigenic Escherichia coli. Infection and Immunity, 75, 1. |
| 181 | oral | 4.00 | *C. hominis*, TU502 | oocysts | human | beta-Poisson | 1.68E+01 | a = 2.7E-01 N50 = 1.68E+01 | diarrhea |
Okhuysen, P. C., Rich, S. M., Chappell, C. L., Grimes, K. A., Widmer, G. ., Feng, X. ., & Tzipori, S. . (2002). Infectivity of a Cryptosporidium parvum Isolate of Cervine Origin for Healthy Adults and Interferon-γ Knockout Mice. Journal of Infectious Diseases, 185, 9. Retrieved from https://academic.oup.com/jid/article/185/9/1320/937719 |
| 183 | oral | 4.00 | Moredun isolate | oocysts | human | beta-Poisson | 4.55E+02 | a = 1.14E-01 N50 = 4.55E+02 | infection |
Blaser, M. J., Duncan, D. J., Warren, G. H., & W-ll, W. . (1983). Experimental Campylobacter jejuni Infection of Adult Mice. Infection and Immunity, 39, 2. |
|
| ||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||

Parameter histogram for exponential model (uncertainty of the parameter)

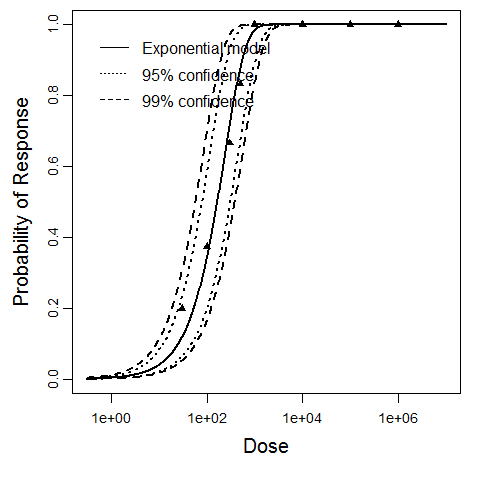
Exponential model plot, with confidence bounds around optimized model
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||

Exponential model plot, with confidence bounds around optimized model

Parameter histogram for exponential model (uncertainty of the parameter)
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||

Parameter scatter plot for beta Poisson model ellipses signify the 0.9, 0.95 and 0.99 confidence of the parameters.

beta Poisson model plot, with confidence bounds around optimized model
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||

Parameter scatter plot for beta Poisson model ellipses signify the 0.9, 0.95 and 0.99 confidence of the parameters.

beta Poisson model plot, with confidence bounds around optimized model.
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||

Parameter scatter plot for beta Poisson model ellipses signify the 0.9, 0.95 and 0.99 confidence of the parameters.

beta Poisson model plot, with confidence bounds around optimized model.