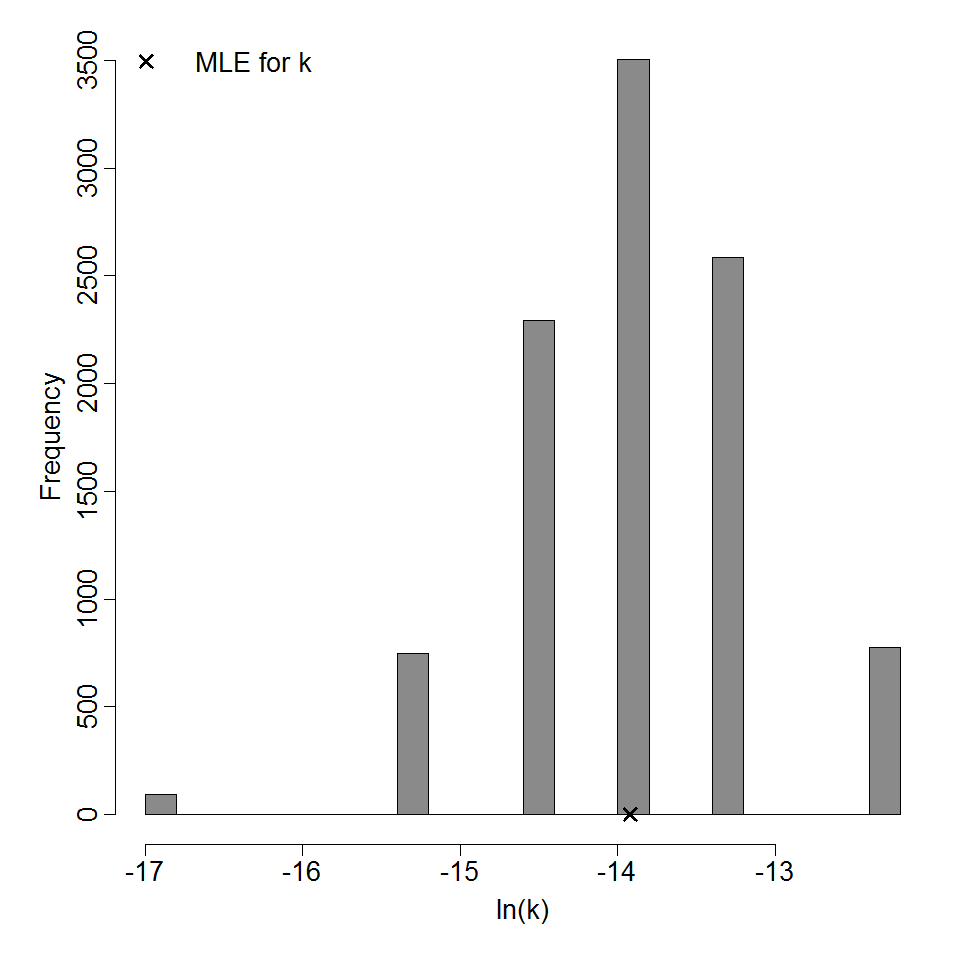General overview
Campylobacter are microaerophilic gram-negative curved or spiral rods with a polar flagellum. C. jejuni and C. coli are common causes of gastroenteritis . It can cause acute self-limiting diarrhea in healthy humans with an incubation period of 2-3 days, and appears very common worldwide. It is mainly a zoonosis, being primarily associated with birds (especially poultry). They do not grow in water and (like Escherichia coli) are an indicator of post-treatment contamination in water distribution systems.
According to feeding studies with chickens, strains of C. jejuni that have been passaged many times in the laboratory tend to have a lower ID50 than strains that are isolated from infected hosts and then used to infect new hosts, with minimal passage . Minimally passaged strains also had more variation in ID50 . Given safety concerns, strains used for human studies may be passaged and studied more, possibly underestimating infectiousness in actual human exposure scenarios .
Campylobacter epidemiology varies greatly between the developed and developing world, probably due to development of immunity early in life. Illness is rare after about 5 years of age (or earlier) in developing countries, but occurs among adults in industrialized countries, probably because they avoided exposure (and therefore immunity) in childhood . However, immunity appears to protect against disease rather than infection, and asymptomatic shedding is common . In a comparison of Mexican children <4 years of age and Swedish patients (ages not given), Swedish patients tended to carry only 1 Campylobacter serotype, while mixed serotypes were common among Mexican children (42%) .
https://www.cdc.gov/campylobacter/
Summary Data
Blaser et al. (1983) experimentally infected adult female HA-ICR mice intragastrically with 3 different serotypes of C. jejuni (strains T1, T2, and T3).
Chen et al. (2006) described dose response models fitted to data from feeding studies in chickens with 19 different strains (18 C. jejuni, 1 C. coli). They found that isolates that had been passaged multiple times in the laboratory were more infectious than isolates taken from infected animals and reused with minimal passaging. Also, one passaged strain (11168) was substantially less infectious than the other passaged strains, which all had very similar dose response curves.
Black et al. (1988) fed human volunteers 2 different strains (81-176 and A3249) of C. jejuni suspended in 150 mL nonfat milk. Neither strain showed an increasing trend of illness with dose. The A3249 strain showed an increasing trend of infection with dose; however, all volunteers became infected with the 81-176 strain regardless of dose. The data describing infection with the A3249 strain were fit by Medema et al. (1996). Teunis et al. (1999) pooled all illness data from Black et al. (1988) to fit a model for strains 81-176 and A3249, which remained similar to Medema et al. (1996). This model was later elaborated by Teunis et al. (2005) by including information from 2 outbreaks of C. jejuni in contaminated milk.
Tribble et al. (2009) fed healthy adult human volunteers with the CG8421 strain of C. jejuni, along with bicarbonate. However, only 2 doses were used, and all but 1 volunteer became ill.
Tribble et al. (2010) experimentally infected humans with strain 81-176 of C. jejuni, one of the strains used by Black et al. (1988) . Although Black et al. (1988) administered the dose in milk, Tribble et al. (2010) administered the dose in a solution containing 2g of bicarbonate. All volunteers were infected, but a dose response trend was seen in the development of disease.
Recommended Model
It is recommended that experiment 106 is preferred in most circumstances. However, consider all available models to decide which one is most appropriate for your analysis.
| ID | Exposure Route | # of Doses | Agent Strain | Dose Units | Host type | Μodel | LD50/ID50 | Optimized parameters | Response type | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| 184 | intragastric | 5.00 | type strain for serotype PEN 1 | CFU | mice | exponential | 7.69E+05 | k = 9.01E-07 | infection |
Blaser, M. J., Duncan, D. J., Warren, G. H., & W-ll, W. . (1983). Experimental Campylobacter jejuni Infection of Adult Mice. Infection and Immunity, 39, 2. |
| 185 | intragastric | 5.00 | type strain for serotype PEN 2 | CFU | mice | beta-Poisson | 6.68E+04 | a = 3.19E-01 N50 = 6.68E+04 | infection |
Blaser, M. J., Duncan, D. J., Warren, G. H., & W-ll, W. . (1983). Experimental Campylobacter jejuni Infection of Adult Mice. Infection and Immunity, 39, 2. |
| 186 | intragastric | 5.00 | type strain for serotype PEN 3 | CFU | mice | beta-Poisson | 3.14E+04 | a = 1.17E-01 N50 = 3.14E+04 | infection |
Blaser, M. J., Duncan, D. J., Warren, G. H., & W-ll, W. . (1983). Experimental Campylobacter jejuni Infection of Adult Mice. Infection and Immunity, 39, 2. |
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
Parameter histogram for exponential model (uncertainty of the parameter)
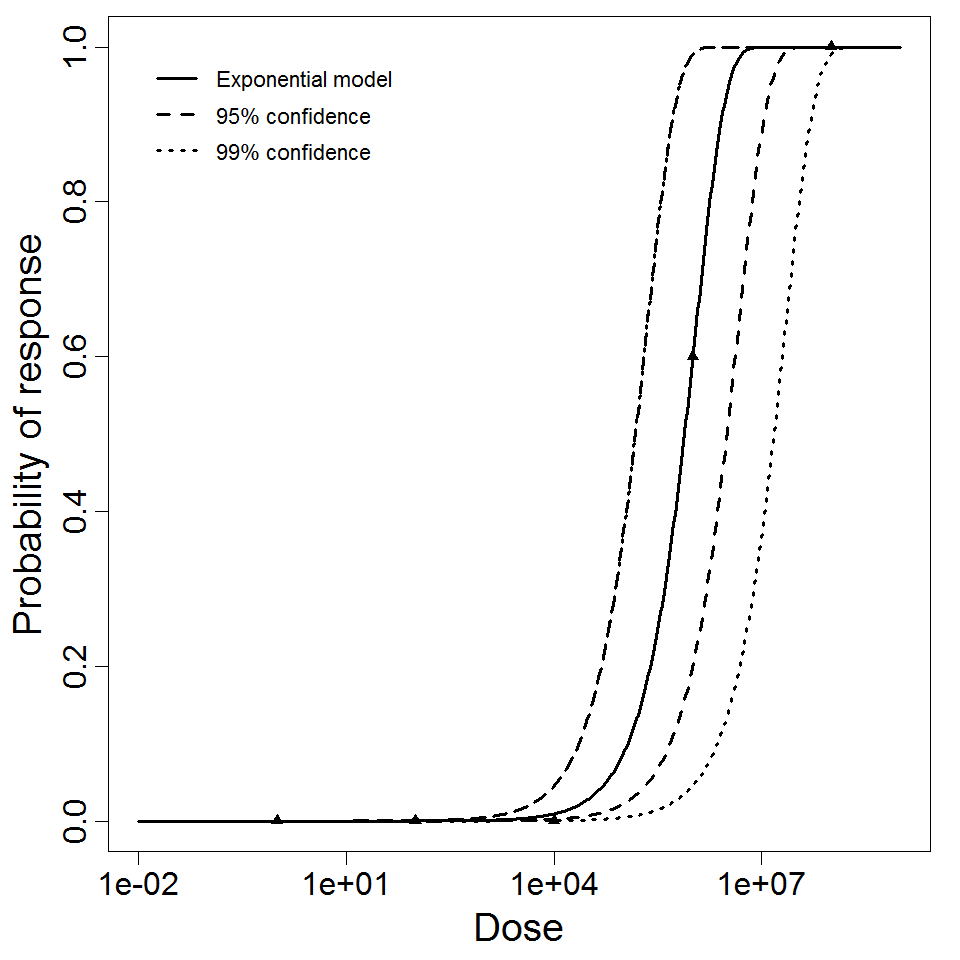
Exponential model plot, with confidence bounds around optimized model
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
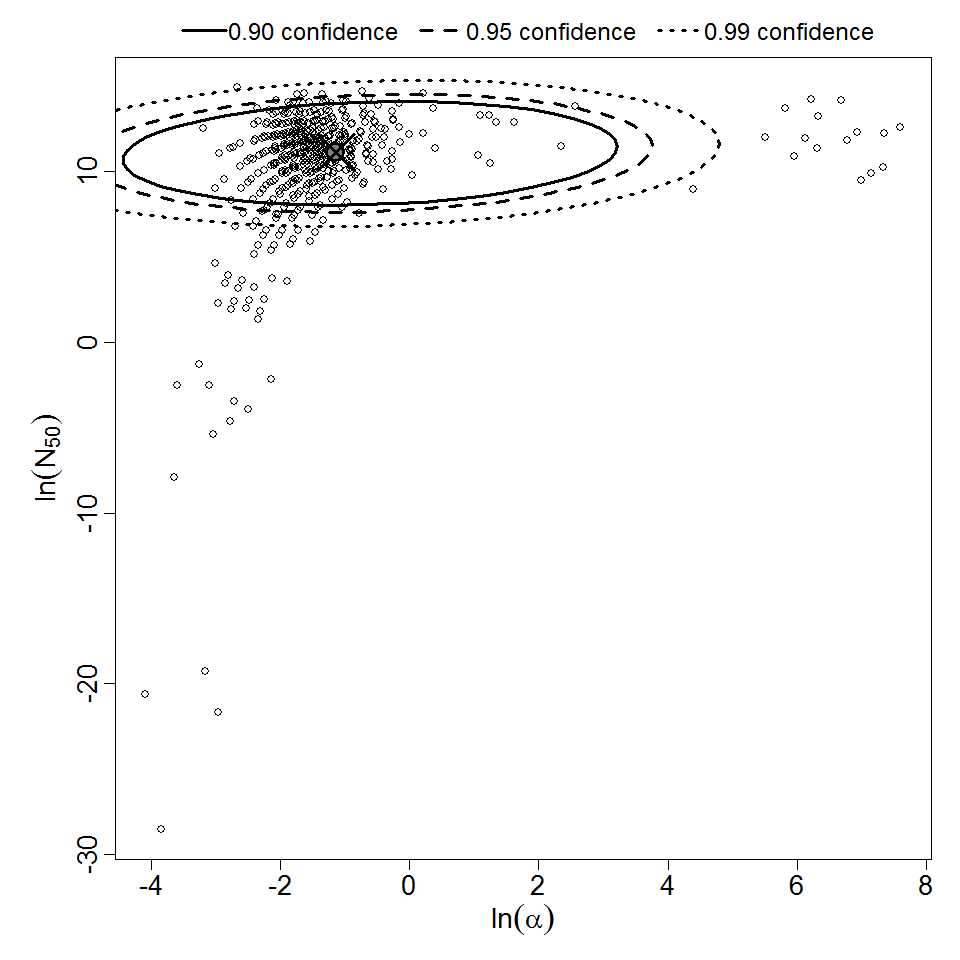
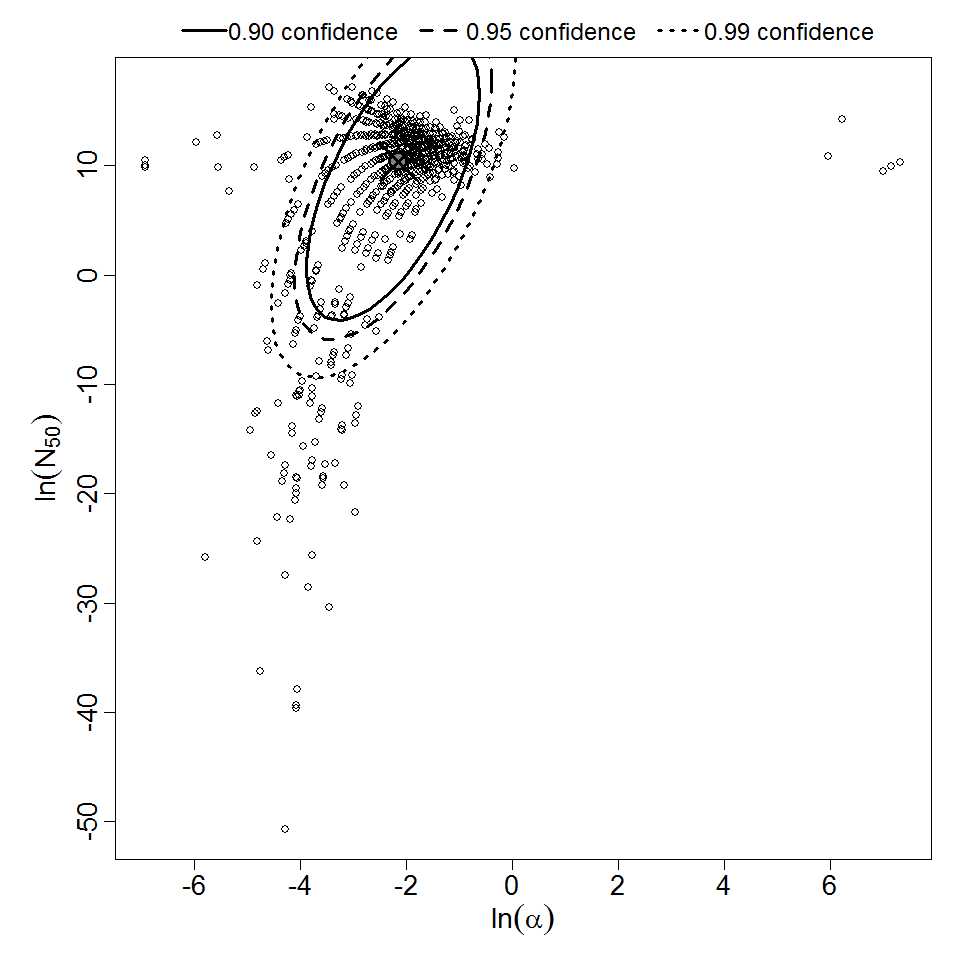
Parameter scatter plot for beta Poisson model ellipses signify the 0.9, 0.95 and 0.99 confidence of the parameters.
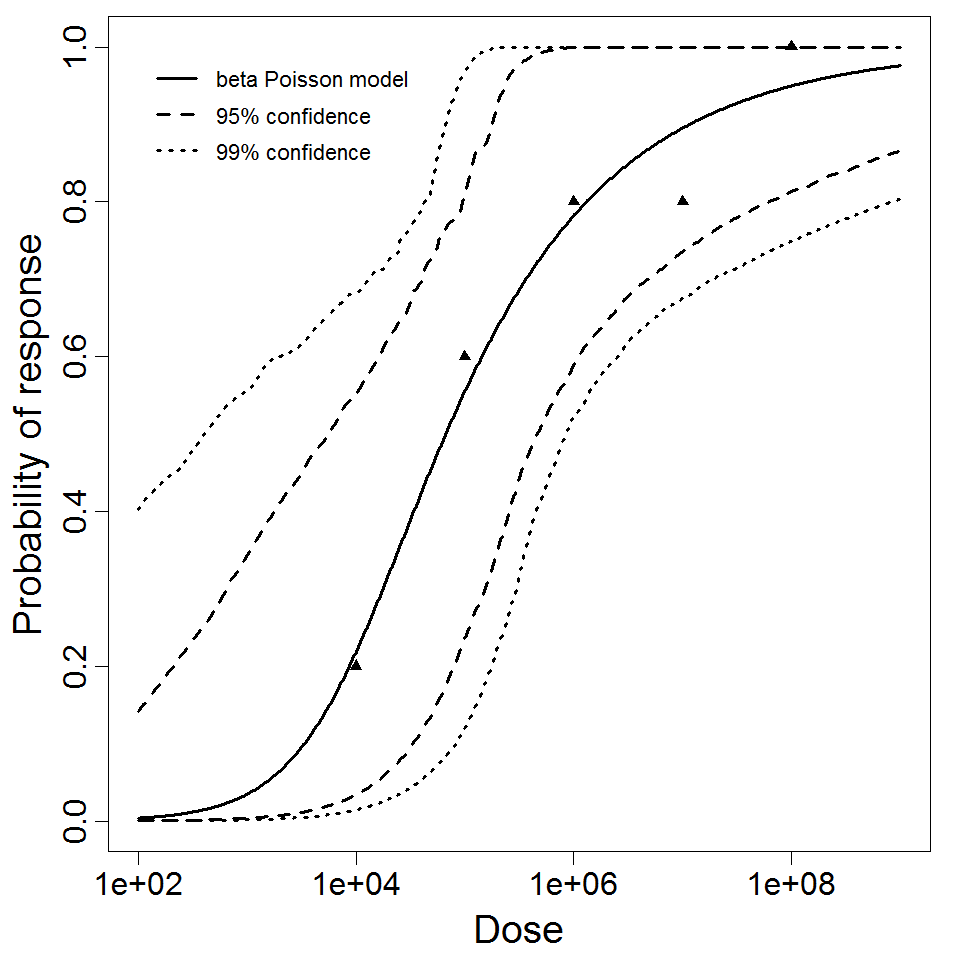
beta Poisson model plot, with confidence bounds around optimized model
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
Parameter scatter plot for beta Poisson model ellipses signify the 0.9, 0.95 and 0.99 confidence of the parameters.
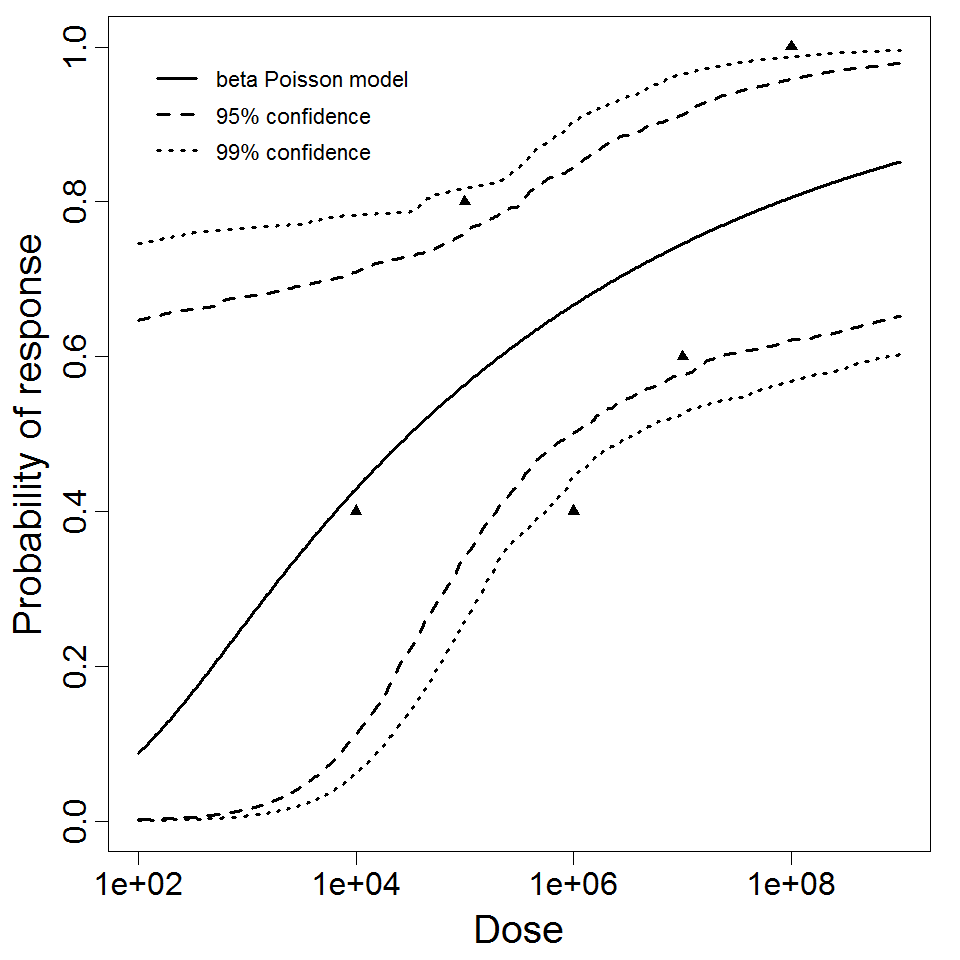
beta Poisson model plot, with confidence bounds around optimized model